
Full article: Urban Oligarchy and Dissolutioned Voters: The End of Monastic Rule in Reading, 1350-1600
![A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials](https://pubs.acs.org/cms/10.1021/cm000804d/asset/images/medium/cm000804df1.gif)
A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials
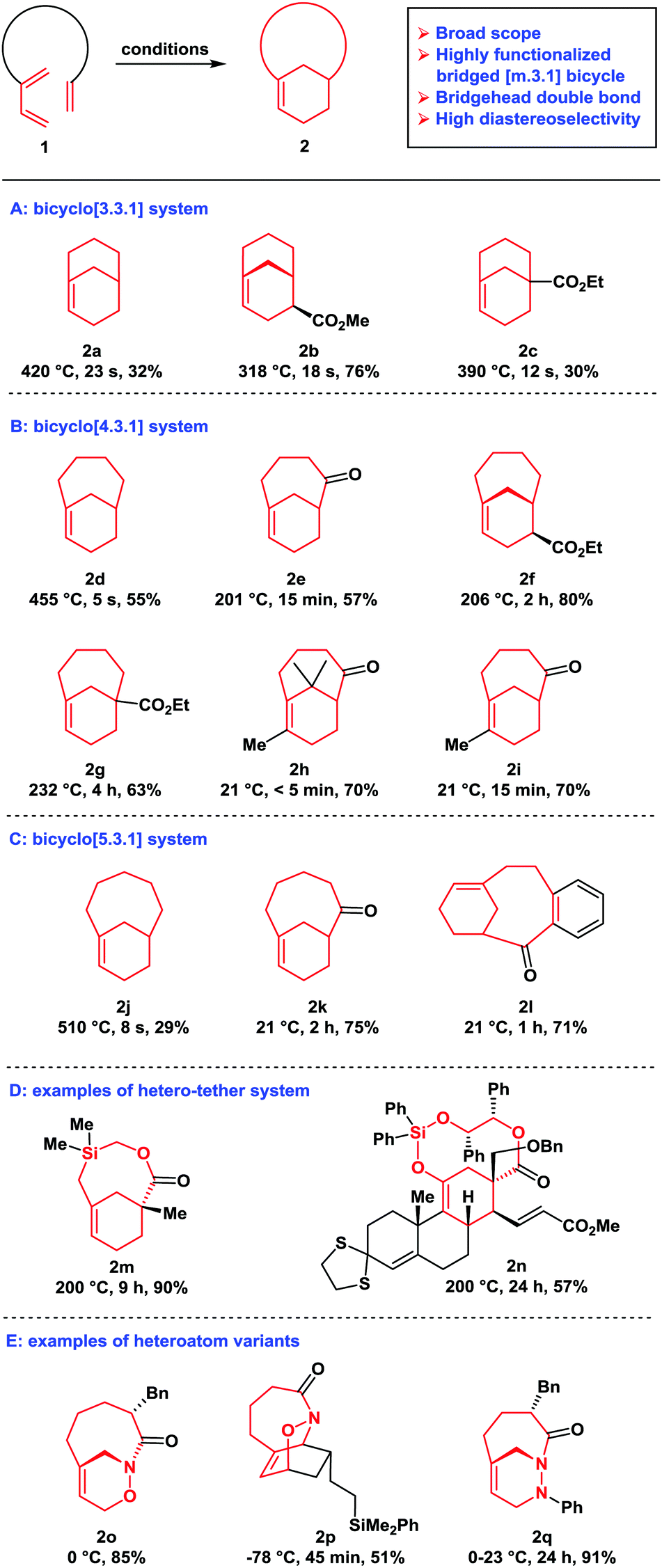
Synthetic applications of type II intramolecular cycloadditions - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D0CS00365D
![A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials](https://pubs.acs.org/cms/10.1021/cm000804d/asset/images/medium/cm000804df00008.gif)
A2[MX4] Copper(II) Pyridinium Salts. From Ionic Liquids to Layered Solids to Liquid Crystals | Chemistry of Materials

On the Release of Hydrogen from the S−H groups in the Formation of Self-Assembled Monolayers of Thiols | Langmuir

Single‐Atom Nanozymes for Biomedical Applications: Recent Advances and Challenges - Tang - 2022 - Chemistry – An Asian Journal - Wiley Online Library
![Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal](https://onlinelibrary.wiley.com/cms/asset/23891b2b-157f-49d7-b63f-4eb2de643e34/ajoc202100523-fig-5001-m.jpg)
Conformational Preferences of Allene Ketones in Lewis Base Catalysis: Synthesis of 4H‐Pyrans and 3,4‐Dihydro‐2H‐pyrans via α‐Regioselective [4+2] Annulations of γ‐Substituted Allene Ketones and Activated Alkenes - Hou - 2021 - Asian Journal
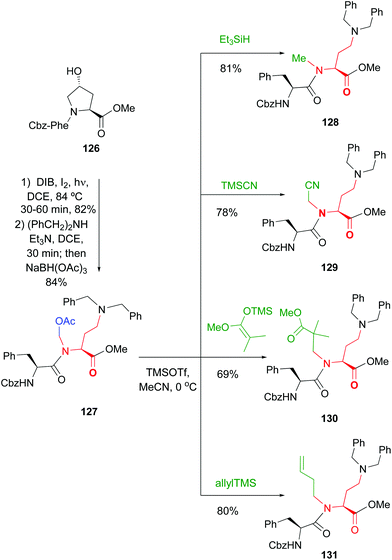
Site-selective modification of peptide backbones - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00892G






![再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS 再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS](https://www.jewels-net.jp/phone/data/jewels/product/4095yndz_1911_1_5.jpg)

![再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS 再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS](https://www.jewels-net.jp/phone/data/jewels/product/4095yndz_1911_1_8_2.jpg)
![再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS 再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS](https://www.jewels-net.jp/phone/data/jewels/product/4095yndz_1911_1_6.jpg)



![再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS 再入荷!刺繍レース/ネックリボン/ラップスカート風/キャバドレス/ミニドレス【XS-Lサイズ/3カラー】[OF08]【YN】dz|即日発送!キャバドレス通販はJEWELS](https://www.jewels-net.jp/phone/data/jewels/product/4095yndz_1911_1_3.jpg)

