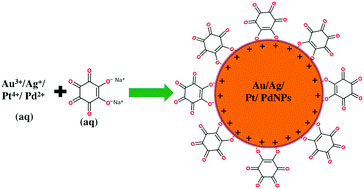
Catalyzed oxidative degradation of methyl orange over Au catalyst prepared by ionic liquid-polymer modified silica

The specialized twin-solution method for selective Pd(II) ions determination and methyl orange removal - ScienceDirect

Rapid Photocatalytic Decolorization of Methyl Orange under Visible Light Using VS4/Carbon Powder Nanocomposites | ACS Sustainable Chemistry & Engineering

Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladium‐Gold Nanoparticles Dendrimer. - Ilunga - 2017 - ChemistrySelect - Wiley Online Library

Degradation of methylene blue and methyl orange by palladium-doped TiO2 photocatalysis for water reuse: Efficiency and degradation pathways - ScienceDirect
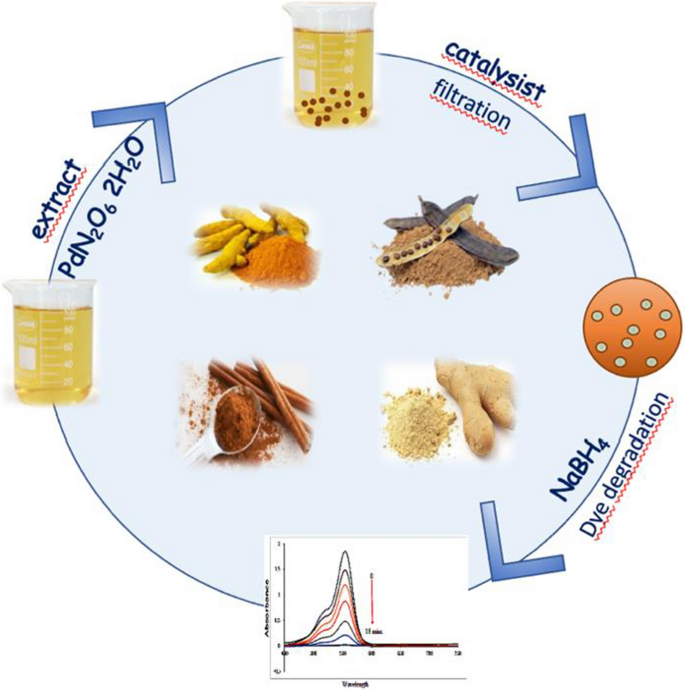
Green synthesis of palladium nanoparticles and investigation of their catalytic activity for methylene blue, methyl orange and rhodamine B degradation by sodium borohydride | SpringerLink

Acceleration of biotic decolorization and partial mineralization of methyl orange by a photo-assisted n-type semiconductor - ScienceDirect

Molecules | Free Full-Text | Synergistic Promotion of Photocatalytic Degradation of Methyl Orange by Fluorine- and Silicon-Doped TiO2/AC Composite Material

Degradation mechanism of Methyl Orange by electrochemical process on RuO(x)-PdO/Ti electrode. | Semantic Scholar

Efficient degradation of methyl orange and methylene blue in aqueous solution using a novel Fenton-like catalyst of CuCo-ZIFs
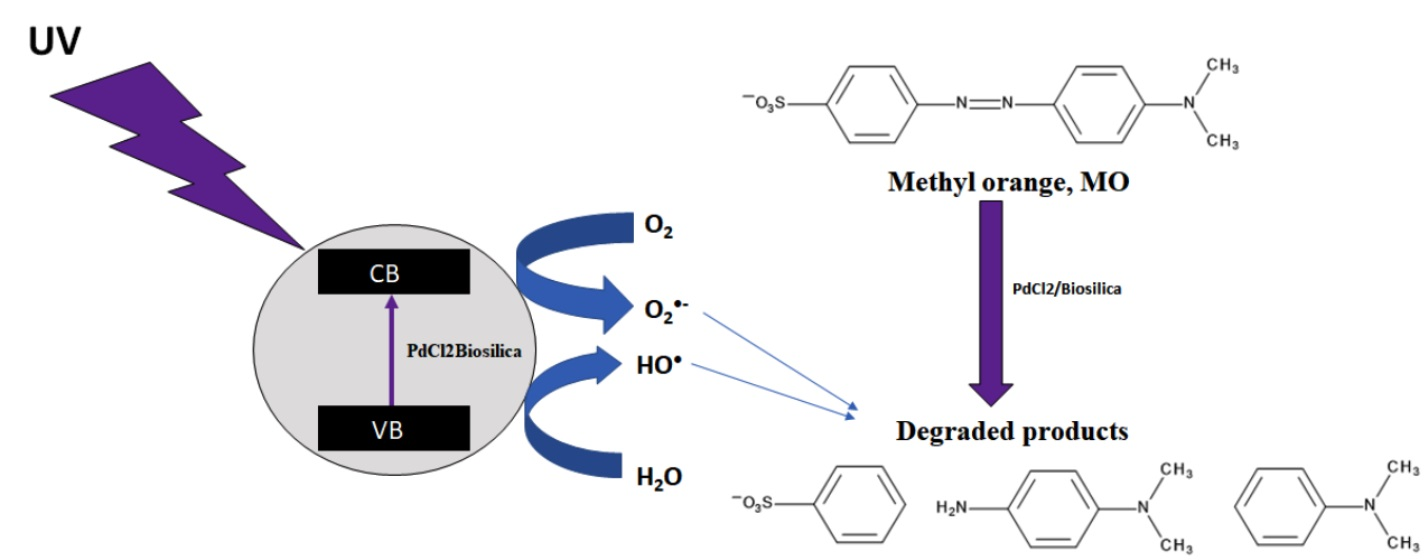
IJMS | Free Full-Text | Diatom Biosilica Doped with Palladium(II) Chloride Nanoparticles as New Efficient Photocatalysts for Methyl Orange Degradation
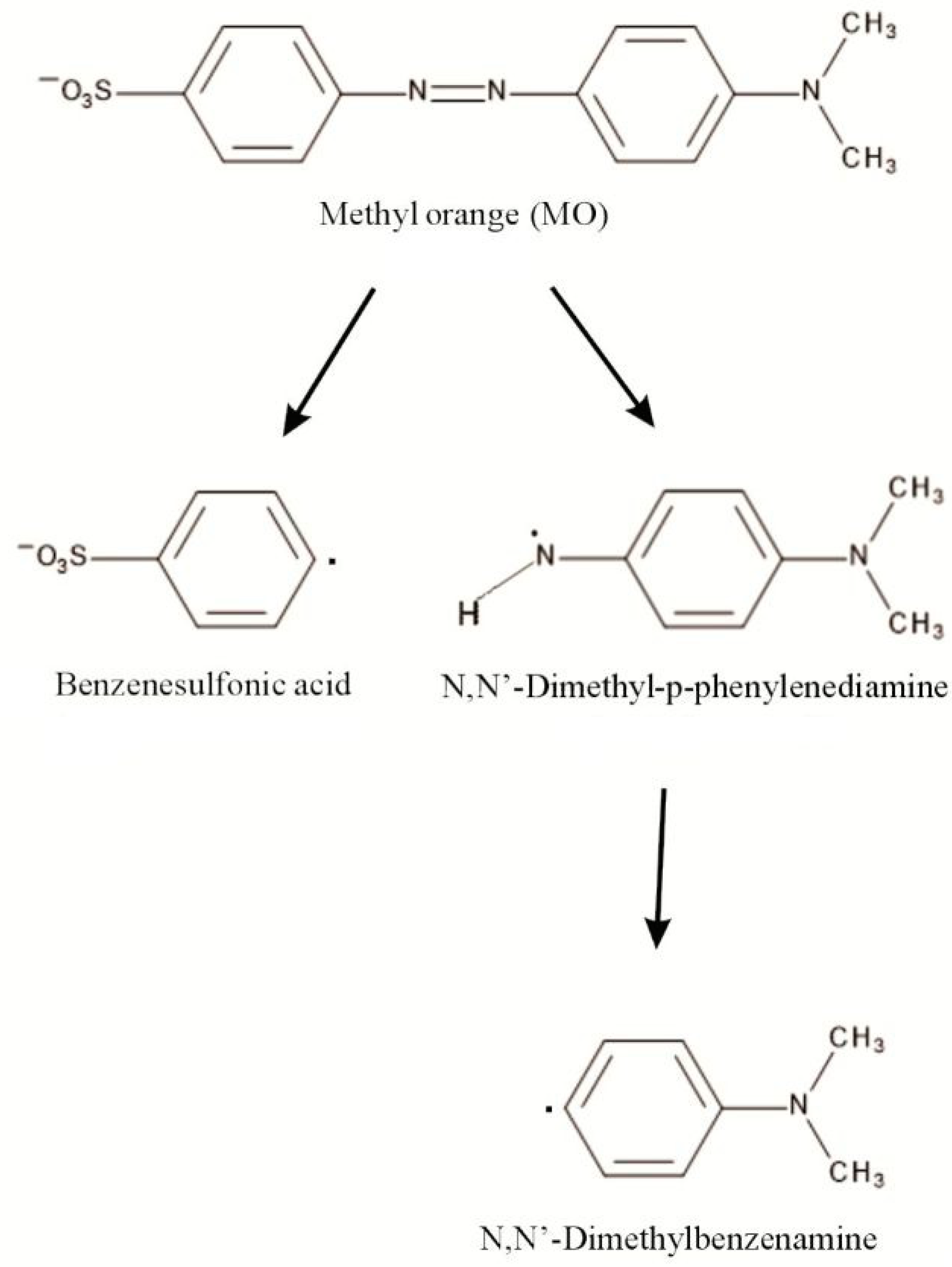
IJMS | Free Full-Text | Diatom Biosilica Doped with Palladium(II) Chloride Nanoparticles as New Efficient Photocatalysts for Methyl Orange Degradation

Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladium‐Gold Nanoparticles Dendrimer. - Ilunga - 2017 - ChemistrySelect - Wiley Online Library

Degradation mechanism and toxicity reduction of methyl orange dye by a newly isolated bacterium Pseudomonas aeruginosa MZ520730 - ScienceDirect

Chemical structure of methyl orange (MO). Linear formula is C14H14N3NaO3S | Download Scientific Diagram

Enhanced Adsorptive Removal of Methyl Orange and Methylene Blue from Aqueous Solution by Alkali-Activated Multiwalled Carbon Nanotubes | ACS Applied Materials & Interfaces
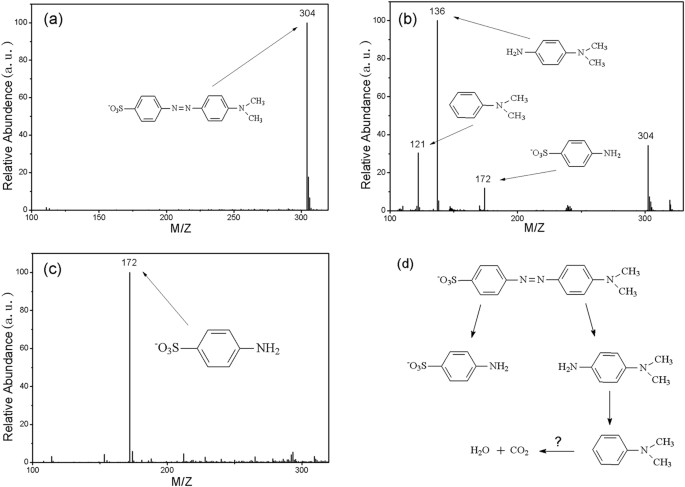
A highly efficient degradation mechanism of methyl orange using Fe-based metallic glass powders | Scientific Reports

Palladium nanoparticles supported on ionic liquid and glucosamine-modified magnetic iron oxide as a catalyst in reduction reactions | SpringerLink

Degradation mechanism of Methyl Orange by electrochemical process on RuO(x)-PdO/Ti electrode. | Semantic Scholar

Pd–Rh Alloyed Nanoparticles on Zeolite Imidazolide Framework-67 for Methyl Orange Degradation | ACS Applied Nano Materials

Pd–Rh Alloyed Nanoparticles on Zeolite Imidazolide Framework-67 for Methyl Orange Degradation | ACS Applied Nano Materials